 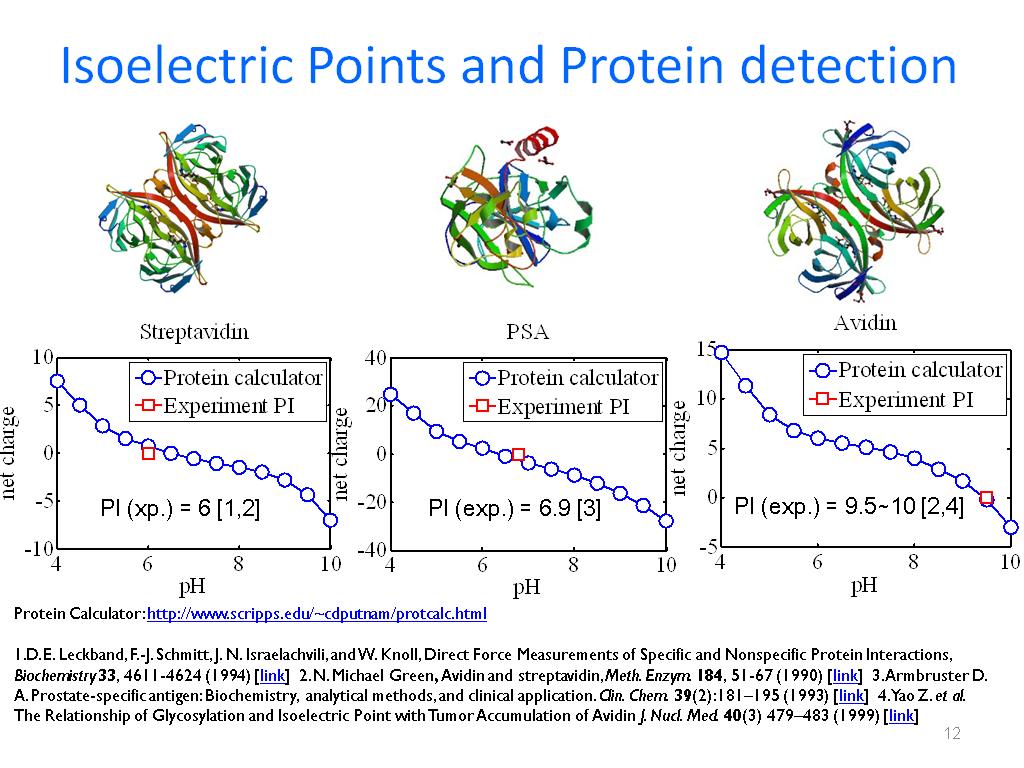
The isoelectric points of 39 Candida parapsilosis strains were determined by means of capillary isoelectric focusing. Ruzicka, Filip Horka, Marie Hola, Veronika Kubesova, Anna Pavlik, Tomas Votava, Miroslav The differences in the isoelectric points of biofilm-positive and biofilm-negative Candida parapsilosis strains. Through this instrument the net charges of the ionizable residues of the chains of the proteins are calculated at different pH values, tabulated, pI is predicted and an Excel… The ability to conduct measurements in free solution thus provides the basis for the rapid determination of isoelectric points of proteins under a wide variety of solution conditions and in small volumes.ĭesign of a Software for Calculating Isoelectric Point of a Polypeptide According to Their Net Charge Using the Graphical Programming Language LabVIEWĮRIC Educational Resources Information CenterĪ software to calculate the net charge and to predict the isoelectric point (pI) of a polypeptide is developed in this work using the graphical programming language LabVIEW. To demonstrate the general approachability of this platform, we have measured the isoelectric points of representative set of seven proteins, bovine serum albumin, β-lactoglobulin, ribonuclease A, ovalbumin, human transferrin, ubiquitin and myoglobin in microlitre sample volumes. 
In particular, in this approach, the pH of the electrolyte solution is modulated in time rather than in space, as in the case for conventional determinations of the isoelectric point. Here, we introduce a gradient-free approach, exploiting a microfluidic platform which allows us to perform rapid pH change on chip and probe the electrophoretic mobility of species in a controlled field. The majority of conventional methods for the determination of the isoelectric point of a molecule rely on the use of spatial gradients in pH, although significant practical challenges are associated with such techniques, notably the difficulty in generating a stable and well controlled pH gradient. The isoelectric point (pI) of a protein is a key characteristic that influences its overall electrostatic behaviour. ÅapiÅ„ska, Urszula Saar, Kadi L Yates, Emma V Herling, Therese W Müller, Thomas Challa, Pavan K Dobson, Christopher M Knowles, Tuomas P J 
Gradient-free determination of isoelectric points of proteins on chip. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
